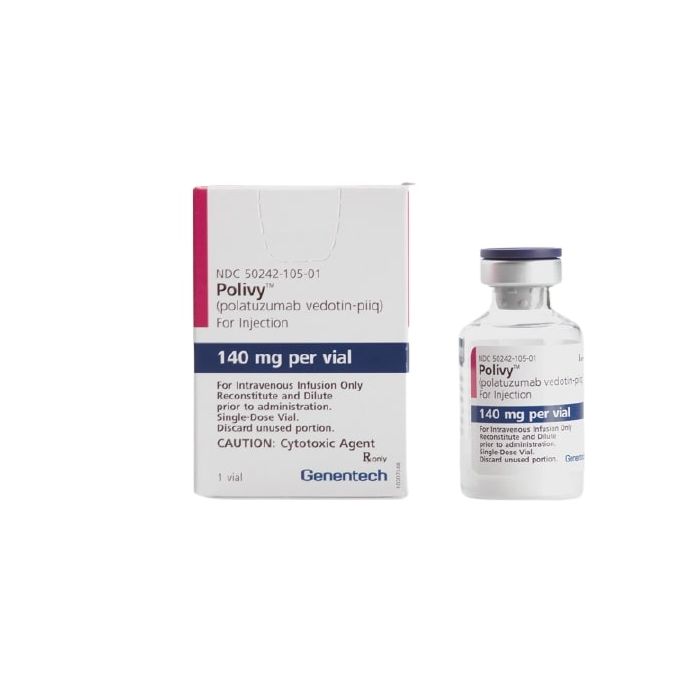
What is Polivy (polatuzumab vedotin-piiq)
Polivy (polatuzumab vedotin-piiq) is a medication indicated in combination with bendamustine and a rituximab product for the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma, not otherwise specified, after at least two prior therapies. Diffuse large B-cell lymphoma is a type of non-Hodgkin lymphoma.
Complete information about Polivy (polatuzumab vedotin-piiq) dosage and administration can be found in the official prescribing information listed in our references section.[1]
Where has Polivy (polatuzumab vedotin-piiq) been approved?
Polivy (polatuzumab vedotin-piiq) has been approved in combination with bendamustine and a rituximab for the treatment of diffuse large B-cell lymphoma by:
- The Food and Drugs Administration (FDA), on June 11, 2019.[1]
- The Pharmaceuticals and Medical Devices Agency (PMDA), Japan on March 23, 2021.
Please note that this medicine may have also been approved in other regions than the ones we’ve listed. If you have a question about its approval in a specific country feel free to contact our support team.
Be the first to review “Polivy (polatuzumab vedotin-piiq) 1 vial of 140 mg” Cancel reply
Related products
OTHER MEDICATIONS
CHEMOTHERAPY DRUGS
OTHER MEDICATIONS
OTHER MEDICATIONS









Reviews
There are no reviews yet.