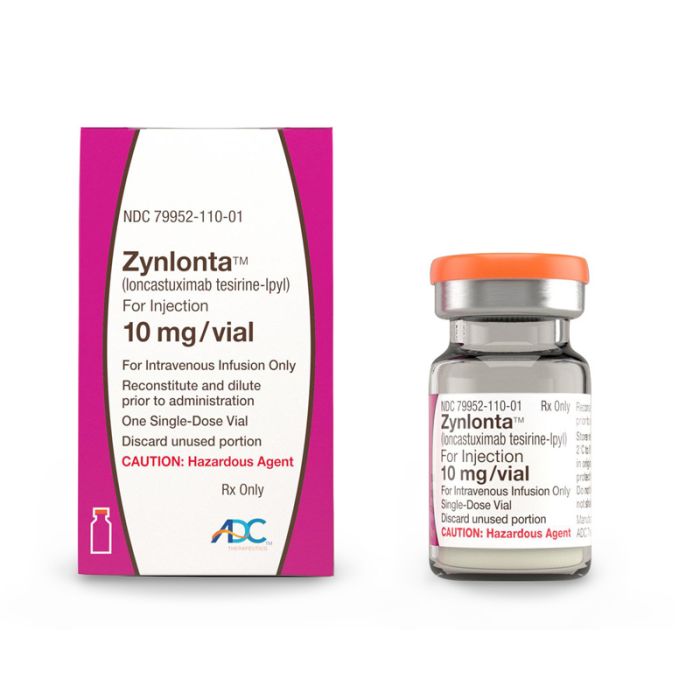
What is Zynlonta (loncastuximab tesirine-lpyl) for?
Zynlonta (loncastuximab tesirine-lpyl) is an antibody-drug conjugate (ADC) used to treat adults with certain types of large B-cell lymphoma that has come back (relapsed) or that did not respond to previous treatment (refractory), who have already received two or more treatments for their cancer. This includes diffuse large B-cell lymphoma (DLBCL) not otherwise specified, DLBCL arising from low grade lymphoma and high-grade B-cell lymphoma.[1]
It is available in vial form for intravenous infusion, containing 10 mg loncastuximab tesirine-lpyl.[1]
How does Zynlonta (loncastuximab tesirine-lpyl) work?
Loncastuximab tesirine-lpylv works differently from chemotherapy or immunotherapy because it is designed to deliver medicine directly to cancer cells.[2,3]
It is an antibody-drug conjugate (ADC) that is made up of the antibody (a type of protein) IgG1 and a cytotoxic (anticancer) compound. IgG1 can bind to CD19 that is present on the surface of cancer cells. As a result, the cancer cell absorbs the medicine, and the drug is released. The cytotoxin can then kill the cancer cell, while leaving the healthy cells intact.[2,3]
Where has Zynlonta (loncastuximab tesirine-lpyl) been approved?
Zynlonta (loncastuximab tesirine-lpyl) was approved for the treatment of adults with advanced large B-cell lymphoma by:
- The Food and Drug Administration (FDA), USA on April 23, 2021.[4]
Zynlonta (loncastuximab tesirine-lpyl) was approved under accelerated approval based on overall response rate.[4]
Please note that this medicine may have also been approved in other regions than the ones we’ve listed. If you have a question about its approval in a specific country feel free to contact our support team.
How is Zynlonta (loncastuximab tesirine-lpyl) taken?
- 0.15 mg/kg body weight every 3 weeks for 2 cycles.
- 0.075 mg/kg body weight every 3 weeks for subsequent cycles.
Zynlonta (loncastuximab tesirine-lpyl) is given as an intravenous (IV) infusion into your vein over 30 minutes. The infusion is given on Day 1 of each cycle (every 3 weeks). The healthcare provider will decide how many treatments are needed.[1]
The treating doctor may give a medicine before each infusion to decrease your chance of side effects.[1]
The treating doctor may stop the treatment, delay treatment, or change the dose if the patient experiences severe side effects.[1]
Complete information about Zynlonta (loncastuximab tesirine-lpyl) dosage and administration can be found in the official prescribing information listed in our references section.[1]
Note: Please consult with your treating doctor for personalised dosing.
Are there any known adverse reactions or side effects of Zynlonta (loncastuximab tesirine-lpyl)?
Common adverse reactions
The most common side effects ( ≥20% of patients) listed in the prescribing information include:[1]
- Feeling tired or weak
- Skin rash
- Swelling
- Nausea
- Muscle or joint pain
- Increase in blood sugar (hyperglycemia)
- Changes in certain blood or laboratory tests
Serious adverse reactions
The serious adverse reactions listed in the prescribing information include:[1]
- Fluid retention
- Low blood cell counts (platelets, red blood cells, and white blood cells)
- Infections
- Skin reactions
Use in a specific population
Zynlonta (loncastuximab tesirine-lpyl) may cause harm to an unborn baby. It is advised to avoid breastfeeding and pregnancies. Females who are able to become pregnant should use effective birth control (contraception) during treatment with Zynlonta and for 9 months after the last dose.
Males who have female partners that are able to become pregnant should use effective contraception during treatment with Zynlonta and for 6 months after the last dose.[1]
For a comprehensive list of side effects and adverse reactions please refer to the official prescribing information.[1]
Be the first to review “Zynlonta (loncastuximab tesirine-lpyl) One vial of 10 mg” Cancel reply
Related products
OTHER MEDICATIONS
OTHER MEDICATIONS
OTHER MEDICATIONS
CHEMOTHERAPY DRUGS
OTHER MEDICATIONS












Reviews
There are no reviews yet.